Former FDA Official Domenic Veneziano Joins Forces with Sandler Travis Law Firm

Former Food and Drug Administration official Domenic J. Veneziano has become FDA regulatory and strategic consultant to Sandler, Travis & Rosenberg.
Pfizer considering request by FDA to change Zoloft warnings
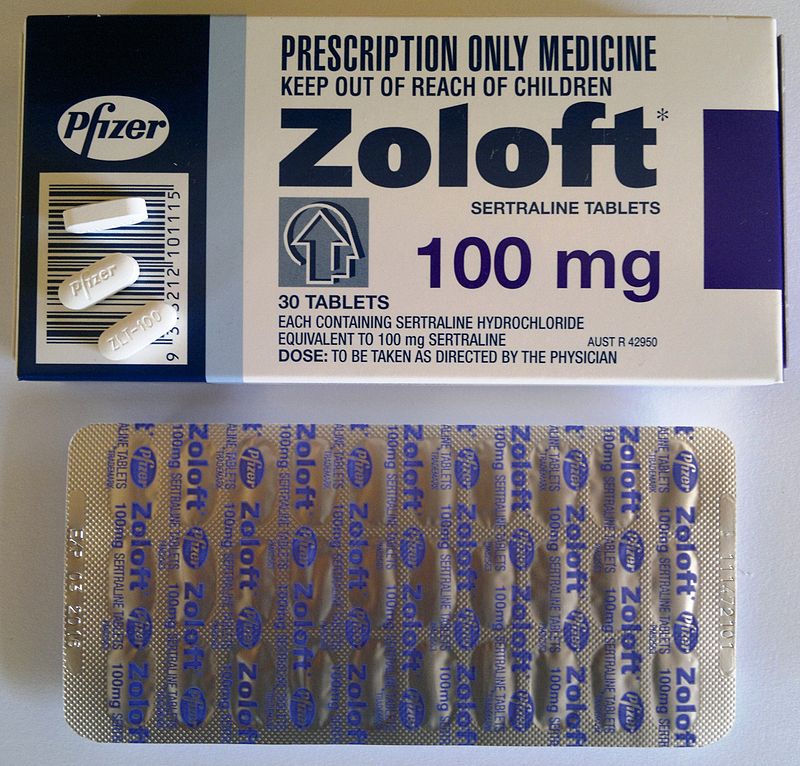
The drug maker is reportedly weighing the request of the FDA to make modifications to its safety warnings for the antidepressant.
New FDA rules make food safety requirements stricter

The FDA has tightened its food safety requirements for manufacturers.
FDA Approves Drug – Treats Rare Metabolic Disorder

The FDA has granted approval for a new drug used to treat hereditary orotic aciduria.
Kim Kardashian under fire for promoting controversial morning sickness drug

The reality star sparked controversy after posting a picture on social media of herself holding a morning sickness medication linked to birth defect fears.
Medtronic recalls over 6,000 units of heart device loading system

The medical device company is recalling thousands of units of the loading system of a heart device recently approved by the FDA.
Toxic dog treats kill more than 1,000 pets

More than 1,000 dogs have died as a result of toxic jerky treats according to the FDA.
States seek reconsideration of new painkiller by FDA

Several states are asking the FDA to reconsider the approval of the new painkiller Zohydro.
Leading Trade and Customs Firm Adds Seasoned Food and Drug Law Counsel
Arnold (Arnie) Friede joins Sandler, Travis & Rosenberg as Of Counsel and Senior Food and Drug Law Attorney
FDA reviews new interpretation of study on Avandia risks
The FDA is set to weigh in on lifting safety restrictions on the diabetes drug Avandia.
Last-minute appeal filed to delay non age restricted morning after pill sales
The government filed an appeal Monday to delay a decision regarding morning after pill sales without prescription to girls without age restrictions.
Morning sickness drug approved for comeback after 30 years
The FDA has approved a drug for morning sickness 30 years after it had been taken off of the market.